
Voltage, Salt Concentration, and Electrolysis Rate
Medium
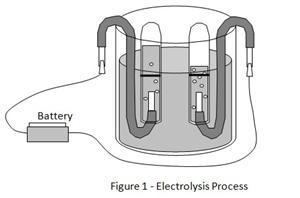
Does more voltage make electrolysis (splitting water with electricity) produce gas faster? You set up two copper wires in a beaker of salt water and connect them to batteries. Gas bubbles form at the wires and rise into upside-down test tubes.
First you test different amounts of salt in the water while keeping the voltage at 1.5 volts. Then you test different voltages while keeping the salt amount the same.
Changing the salt amount barely affects the speed. But raising the voltage from 1.5V to 6V cuts the collection time from about 14 minutes down to under 5 minutes.
Hypothesis
The hypothesis is that a higher DC voltage applied to the electrolysis process will increase the rate at which hydrogen gas is produced.
Science Concepts Learned
Electrolysis
Electrolysis splits water with electricity — and voltage controls how fast that splitting happens. When you raise the voltage from 1.5V to 6V, the collection time drops from about 14 minutes down to under 5 minutes.
Hydrogen Gas
Hydrogen is the lightest gas, so it rises fast when freed from water by electricity. Two copper wires in a beaker of salt water carry current that splits the water, and gas bubbles form at the wires and rise into upside-down test tubes. Changing the salt amount barely affects the speed. But raising the voltage from 1.5V to 6V cuts the collection time from about 14 minutes down to under 5 minutes.
Method & Materials
You will use a beaker, tap water, salt, batteries, test tubes, and other materials to conduct the experiment.
You will need a beaker, tap water, salt, batteries, test tubes, and other materials.
MEL Chemistry — hands-on chemistry experiment kits delivered monthly — great for building lab skills at home. (Affiliate link)
See what’s includedResults
The results of this experiment show that increasing the DC voltage will result in a faster release of gasses from the chemical reaction in the electrolyte. However, increasing the amount of salt in the electrolyte does not change the rate of gas released from the electrolysis process.
Why do this project?
This science project is interesting because it explores the process of electrolysis and how electricity can be used to create gas.
Also Consider
Consider experimenting with different types of electrolytes such as vinegar or sulfuric acid. Also, examine the effects of raising or lowering the temperature of the solution during the process of electrolysis.
Full project details
Additional information and source material for this project are available below.Related videos
These videos explain the science behind this project and demonstrate key concepts used in the experiment.
Share this Science Project:
Related Science Fair Project Ideas
Add Alka-Seltzer to a red cabbage lava lamp and watch the blobs shift color as you change the pH.
Medium
Grow sparkling rainbow crystals overnight on pipe cleaners and watch solid crystal structures form as the Borax water cools.
Medium
Turn a hollow eggshell into a miniature geode by growing Borax crystals inside it overnight.
Medium
Share this Science Project:
