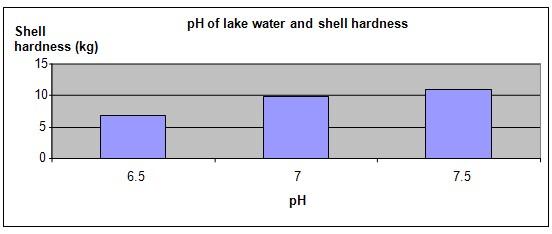
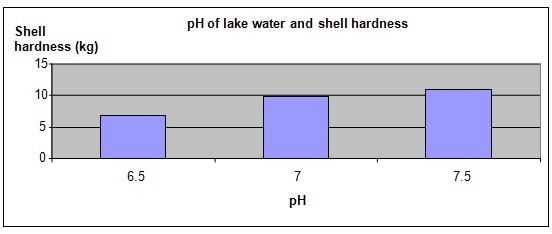
Hypothesis
A lake with more acidic water will produce Zebra mussels with softer shells.
Overview
Zebra Mussels
Zebra mussels belong to a family of clams that have hard, elongated and asymmetrical shells. They are marked by alternating light and dark bands and normally live in slow moving or still fresh water. Zebra mussels will locate any hard surface under water and attach themselves to it. They are often found attached to rocks, buoys, boat hulls and submerged woods.
Zebra mussels usually require a small concentration of calcium in the water to grow their shells. They grow best and most quickly in waters of pH values between 7.4 and 8.4. Waters with pH levels below 6.8 is thought not to be conducive for their growth because the acidic waters may dissolve the shell. Although Zebra mussels can survive for a short while at temperatures below freezing, they thrive at temperatures between 20°C to 25°C. Apart from temperature, Zebra mussels are also sensitive to chemicals that are dissolved in the water and they can also detect touch and gravity.
Zebra mussels are filter feeders. They draw water into their incurrent siphon and then into the branchial chambers where their gills filter food from the water. The waste water then exit through the excurrent siphon. Their diet consists mainly of single-cell organisms such as bacteria, protozoa, plankton and algae.
There are many factors that determine the acidity of a lake, such as the amount of acid rain that the lake receives, whether livestock is found nearby, and whether nitrogen-based fertilizers are used in the vicinity. Also, the soil type in the surrounding catchment area plays a part in determining the acidity of lake water. If the soil is very alkaline in nature, it will neutralize the acid rain as the rain water seeps through the soil, on its way into the lake.
Scientific Terms
Zebra mussels, clams, asymmetry, pH, siphon, branchial chambers, gills
Conclusion
The hypothesis that a lake with more acidic water will produce Zebra mussels with softer shells is proven correct.
Pollutant gasses released into the atmosphere by factories and vehicles return to us in the form of acid rain. The acidification of our oceans and fresh water lakes threatens the survival of plants, animals and fish that live in the water. Crustaceans and clams, are now more vulnerable because their shells, which are the only protection that they have, are slowly dissolving and weakening as a result of the acidification of water bodies such as lakes and oceans.
Also consider
What would happen if this science fair project were to be repeated with seawater mussels instead of freshwater mussels?
Would your results differ if a larger sample were to e used? (ie: samples of mussel shells from 20 different lakes, of varying pH levels?) Would the results be perhaps more accurate, given the larger sample?
You should also consider checking calcium levels in the water to ascertain if this makes a difference to your results.
References
Mussel - http://en.wikipedia.org/wiki/Mussel
Zebra mussel - http://animaldiversity.ummz.umich.edu/site/accounts/information/Dreissena_polymorpha.html
Related videos
These videos explain the science behind this project and demonstrate key concepts used in the experiment.