| Complexity level: | 8 |
| Project cost ($): | 30 |
| Time required: | 1 hour to prepare, 1 hour for experiment |
| Material availability: | May be purchased from a hobby store, or alternatively obtained from a school science laboratory |
| Safety concerns: |
Hypothesis
Sea water with higher salinity will conduct more electricity.
Overview
Electrolysis process
Electrolysis is the process where electric current passes through a solution. The process requires a voltage source, an anode (positive) electrode, a cathode (negative) electrode and an electrolyte solution.
The electrolysis process will start when the positive terminal of a battery is connected to the anode and the negative terminal of the battery is connected to the cathode. When this connection is done, the positive ions in the electrolyte will move towards the cathode and the negative ions will move towards the anode resulting in current flow.
Water by itself is a poor conductor. But if salt or sodium chloride is added to water, it can start to conduct electricity through the electrolysis process.
Sodium chloride in solid form does not conduct electricity. The electrostatic attraction between the sodium ion and chloride ion is very strong when it is in solid form. However when sodium chloride is mixed in water, the sodium and chloride ions are separated and move freely in the solution making it a good electrolyte.
Scientific Terms
Materials
The materials required for this science fair project:
- 2 beakers
- 300ml distilled water
- 300ml sea water
- 2 copper electrodes (either a copper rod, copper bar or copper wire)
- 4 1.5V batteries
- Battery holder
- 4 jumper wires with crocodile clips at both ends
- A digital voltmeter
- An ammeter
- A small light bulb with socket
MEL Physics — hands-on physics experiment kits delivered monthly — real experiments, not just reading. (Affiliate link)
See what’s includedProcedure
1. For this experiment, the independent variable is the use of seawater or distilled water. The dependent variable is the amount of current flowing in the circuit as indicated by the bulb lighting up, and the brightness of the bulb. This is measured by using the ammeter to measure the current and observing the light bulb. The constants (control variables) are the battery voltage, light bulb wattage and the amount of electrolyte solution used.
2. Label the beakers as A and B. Fill beaker A with 300ml of distilled water and beaker B with 300ml of salt water.
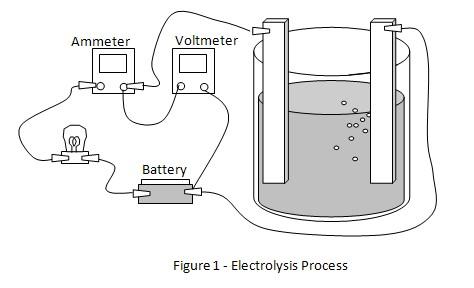
3. Place both copper electrodes in beaker A. The electrodes must not touch one another. Connect one jumper wire from the battery to one of the copper electrodes. The other wire from the battery is connected through the light bulb and ammeter to the second electrode in the beaker. Once the connection is done, the electrolysis process will begin. Please refer to figure 1 for the circuit connection. The voltage can be measured using the digital voltmeter between the electrodes.
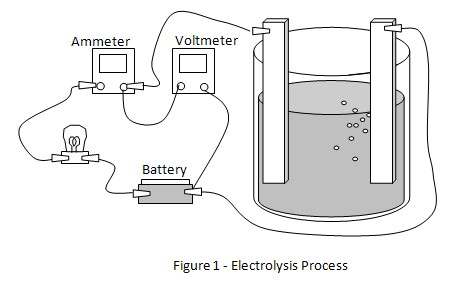
4. Observe the light bulb, ammeter readings and voltmeter readings and record the observations in the table given below.
5. Repeat procedures 3 and 4 using the seawater in beaker B and record the observation and readings in the table below.
Results
The results show that the bulb will light up when sea water is used but does not light up when distilled water is used. The current measured was also higher for sea water. However the voltage measured was almost the same for both electrolyte solutions.
|
Electrolyte Solution |
Voltmeter (V) |
Ammeter (A) |
Light bulb |
|
Distilled water |
5.95 |
0.01 |
Not lighted |
|
Sea water |
5.85 |
0.12 |
Lighted up |
Conclusion
The hypothesis that sea water with a higher salinity will conduct more electricity is proven to be true. The presence of salt in seawater makes it a good conductor.
Electrolysis process can be used to store energy and reproduce it for later use. Examples are the rechargeable and disposable batteries and batteries used to start cars. Currently environment friendly energy sources like solar panels and wind turbines make use of batteries to store the energy that they produce for later use.
Also consider
The experiment can also be done by varying the amount of salt in the water.
This experiment can also be repeated to observe the effect of different sizes and types of electrodes.
References
Electrolysis - http://en.wikipedia.org/wiki/Electrolysis
Electrolysis - http://www.tutorvista.com/content/chemistry/chemistry-iii/redox-reactions/electrolysis.php
Electrolysis - http://www.educationalelectronicsusa.com/c/electrolysis-I.htm

