| Complexity level: | 6 |
| Project cost ($): | 20 |
| Time required: | 1 hour to prepare, 1 hour for science fair project |
| Material availability: | A car with leather seats and another with fabric seats, are required |
| Safety concerns: |
Hypothesis
Leather car seats produce more static electricity compared to fabric seats.
Overview
Static Electricity
The accumulation of static charges on the surface of a body creates static electricity. The electric charges will be discharged when contact is made with another surface.
All matter consists of atoms. The atom has a nucleus at its core, which consists of neutrons and protons while electrons are located outside the nucleus. The tribo-electric series ranks the ability of an atom to retain or release its electron.
An atom that releases its electrons and becomes positively charged will be located on the positive side of the tribo-electric series whereas an atom accepting an electron will be negatively charged and will be located on the negative side of the tribo-electric series.
The process of rubbing 2 surfaces of insulating materials produces static charges. The rubbing action creates friction between the materials which will aid in the generation of static electricity. This will result in electrons being transferred from the atom, releasing electrons to the material containing the atom that receives the electron.
Scientific Terms
Materials
The materials required for this science fair project experiment:
- 1 balloon
- 1 copper plate 200mm x 300mm
- 1 ground connection
- 1 jumper wire with crocodile clips at both ends
- 1 fabric car seat
- 1 leather car seat
- 1 polyester cloth
- 1 cotton cloth
- 1 nylon cloth
- 1 silk cloth
- a flat tray
- 1 ruler
- 1 large sheet of paper
- 1 paper knife
MEL Physics — hands-on physics experiment kits delivered monthly — real experiments, not just reading. (Affiliate link)
See what’s includedProcedure
1. For this science fair project, the independent variable is the type of materials tested – namely fabric and leather car seats against cotton, polyester, nylon and silk cloths. The dependent variable is the number of paper pieces picked up by the balloon. The constants (control variables) are the size of the balloon, the number of times the balloon is rubbed on the material tested, the direction of rubbing, and the weight of the paper bits.
2. The copper plate is connected to the ground connection using the jumper wire.
3. 200 pieces of paper measuring 5mm by 5mm are cut out from the same sheet of paper. The pieces of paper are discharged from all electrical charges, by placing them on the grounded copper plate. They are then transferred to the flat tray.
4. The inflated balloon is discharged by rolling it over the grounded copper plate.
5. To start the science fair project, rub the balloon with the cotton cloth 5 times. Place the cut pieces of paper on the cloth car seat. The paper pieces will be attracted to the balloon. Count the number of paper pieces sticking to the balloon and record the quantity in the table given below. Discharge the paper pieces on the copper plate and return them to the tray.
6. Discharge the balloon again by rolling it on the copper plate. Repeat procedure 5 by placing the cut pieces of paper on the leather car seat. Record the number of papers collected .
7. Repeat procedures 5 and 6 by rubbing the balloon 5 times over the polyester cloth, nylon cloth and silk cloth in turn. Count the number of paper pieces collected and record your findings.
Results
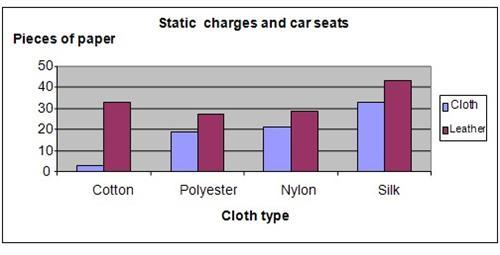
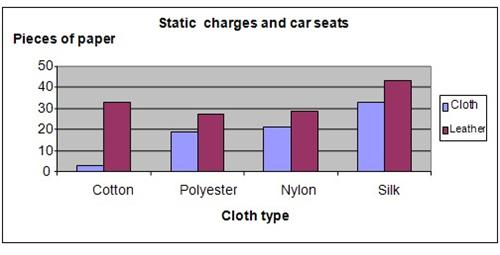
The results showed that the car seat made of fabric or covered with cotton shirts produced the least amount of static electricity. The leather car seat produced more static compared to the fabric seats.
|
Car seat material |
Number of paper pieces collected |
|||
|
Cotton |
Polyester |
Nylon |
Silk |
|
|
Cloth |
3 |
19 |
21 |
33 |
|
Leather |
33 |
27 |
29 |
43 |
The chart below summarizes our findings.
Conclusion
The hypothesis that leather car seats produces more static electricity compared to fabric seats is proven to be true.
The static charge present on surfaces is higher during periods of low humidity. They pose a great danger at gas stations where flammable gases are constantly present. A sudden spark caused by the rubbing of a person’s clothing against the car seat while the person gets out of the car could be extremely disastrous.
Also consider
The science fair project can perhaps be repeated using a comb instead of a balloon.
Would different car seat materials (eg. seats made from cloth or Vinyl or PVC) produce different results?Reference:
References
Static electricity - http://en.wikipedia.org/wiki/Static_electricity
Static electricity - http://science.howstuffworks.com/vdg1.htm

