| Complexity level: | 8 |
| Project cost ($): | 20 |
| Time required: | 1 hour to prepare, 1 hour for the science project experiment |
| Material availability: | Access to basic laboratory equipment required |
| Safety concerns: |
Hypothesis
The peroxidase enzyme will become ineffective as a catalyst at high temperatures.
Overview
The enzyme Peroxidase
The chemical processes that take place in cells are controlled by certain catalysts called enzymes. These enzymes are made from proteins. They help to reduce the amount of energy required to activate certain chemical processes. They do so, by binding with a substrate to produce the end products. The reduction in the required energy to activate the chemical process makes the chemical reactions happen more quickly.
The enzyme called peroxidase is one of the most common and important enzymes found in almost all living organisms. This enzyme is similar to another enzyme called catalase, but peroxidase has a slightly higher temperature tolerance. The enzymes peroxidase and catalse help to convert hydrogen peroxide into water and oxygen.
Hydrogen peroxide is a byproduct of the chemical reactions taking place in cells and is toxic to cell structures. With the help of the enzymes peroxidase and catalase, the toxic hydrogen peroxide is converted into harmless oxygen and water.
Scientific Terms
Materials
The materials required for this science fair project:
- 1 potato
- 7 beakers
- 7 test tubes
- 1 hot plate
- 1 packet of ice
- 1 bottle of hydrogen peroxide
- 1 stop watch
- 1 thermometer
- Marker pen
Eureka Crate — engineering & invention kits for ages 12+ — monthly projects that build real-world skills. (Affiliate link)
See what’s includedProcedure
1. For this experiment, the independent variable is the temperature of the potato and hydrogen peroxide. The dependent variable is the reaction in the test tube. This is determined by counting the number of bubbles formed in the test tube. The constants (control variables) are the amount of hydrogen peroxide in the test tube and the size of the cut potato.
2. The potato is cut into 8 small cubes of 5mm x 5mm x 5mm.
3. The 8 beakers and 8 test tubes are marked 1 to 8 with the marker pen. The test tubes are each marked with a line 20mm from the bottom of the tube. The hydrogen peroxide solution is poured into the 8 test tubes up to the level of the line.
4. The 8 beakers are filled with water up to 6cm high. By adding some ice to the water, the temperature of the first beaker 1 is brought to 15°C. Using the hot plate, the temperatures of the water in beakers 2 to 8 are brought to 25°C, 35°C, 45°C, 55°C, 65°C and 75°C. Place the test tubes marked 1 to 8 into the beakers marked 1 to 8 accordingly. At the same time also put one piece of cut potato into each beaker. Allow the test tubes and the potato’s temperature to stabilize in the water for 5 minutes. Measure the temperature of each tube using the thermometer.
5. Put the potato from the 1st beaker into the test tube of the same beaker. Start the stopwatch and count the number of bubbles released in the next 20 seconds. Record the measurements in the table below.
6. Repeat procedure 5 for the remaining 7 beakers and record all the results in the table below.
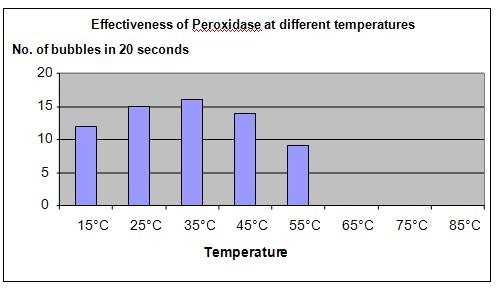
Results
It was observed that bubbles were formed in test tubes 1 to 5. No bubbles were formed in test tubes 6, 7 and 8.
|
Test tube number |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
|
Temperature |
15°C |
25°C |
35°C |
45°C |
55°C |
65°C |
75°C |
85°C |
|
Number of bubbles seen in 20 seconds |
12 |
15 |
16 |
14 |
9 |
0 |
0 |
0 |
The graph below represents the results of our experiment.
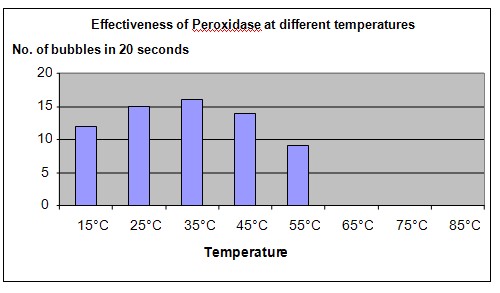
Conclusion
The hypothesis that the enzyme peroxidase will become ineffective as a catalyst at high temperatures is proven to be true. The enzyme is made of protein which will become unstable at higher temperatures. At temperatures above 55°C, the protein in the enzyme peroxidase becomes thermally "denatured" and can longer combine with substrates effectively.
The peroxidase enzyme plays a very important role in reducing the toxin hydrogen peroxide which is formed in cells, into water and oxygen. Peroxidase also has industrial applications. It is used in industrial waste water treatment to remove pollutants.
Also consider
What would happen if the experiment was repeated using different amounts of hydrogen peroxide in the test tubes?
The experiment can be repeated using animal liver tissue instead of potato.
References
Enzyme - http://en.wikipedia.org/wiki/Enzyme
Peroxidase - http://en.wikipedia.org/wiki/Peroxidase