| Complexity level: | 5 |
| Time required: | 15 minutes to simmer fruit. 1-2 hours for set-up and completion of other lab steps. |
| Material availability: | Most materials can be found at a grocery store. Iodine can be purchased from a drug store or online. Graduated cylinders and lab hot plates can likely be found in a school laboratory or at laboratory supply store. |
| Safety concerns: |
Hypothesis
Overview
Vitamin C is a critical nutrient found in a variety of fruits and vegetables. Most commonly, we think of citrus fruits as great sources of Vitamin C ? oranges, lemons, grapefruits and limes ? but other foods are also full of Vitamin C. These include strawberries, cantaloupe and even sweet peppers.
Without enough Vitamin C, we would experience a vitamin deficiency that could lead to damaging health effects. One of the worst illnesses caused by Vitamin C deficiency is scurvy. This disease was common among seafarers during the 1800s and early 1900s; as a result, officials required sailors to consume a citrus ration of limes or lemons. Scurvy victims heal more slowly than healthy individuals, and their gums may begin to bleed. People with prolonged Vitamin C deficiencies may even suffer from slower bone growth.
Vitamins and minerals can easily be destroyed by the cooking process, though some foods actually become more nutritious when heated up. Did you know that cooking foods such as carrots, spinach, peppers and mushrooms may actually help humans absorb more nutrients as opposed to b eating them raw? Today, we will try to determine whether cooking actually increases the amount of available Vitamin C in two foods: tomatoes and cherries. We will find out whether cooked fruits and vegetables are more nutritious (in terms of Vitamin C) than raw fruits.
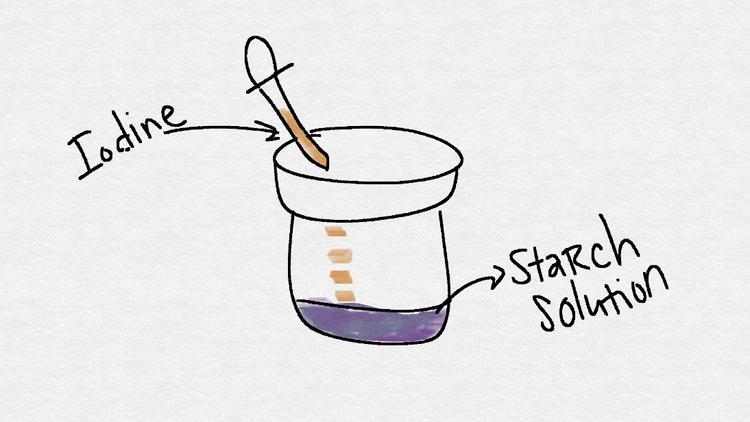
We know that iodine reacts with Vitamin C, so we will be adding it to raw fruit and vegetable extracts and then repeating the experiment with cooked food. Iodine solutions in water and starch lighten when they are exposed to large amounts of Vitamin C. That is, a purple iodine solution will get lighter if Vitamin C is present. This science project involves a titration test to determine the amount of Vitamin C in each sample.
Scientific Terms
Materials
- 1 cup raw cherries (pits and stems removed)
- 1 cup chopped raw tomatoes
- Food processor
- Coffee filters
- 10 mason jars with at least 250 mL capacity
- Laboratory hot plate
- 2% iodine solution
- 5 eyedroppers
- Cornstarch
- Distilled water
- Graduated cylinder
- Funnel
- Permanent marker
- Plastic spoons (can rinse and re-use)
MEL Chemistry — hands-on chemistry experiment kits delivered monthly — great for building lab skills at home. (Affiliate link)
See what’s includedProcedure
- Take the cup of chopped tomatoes and put them in the food processor.
- Blend the tomatoes until they have the consistency of a puree.
- Turn the machine off.
- Using the graduated cylinder, add 150 mL of water into the food processor.
- Blend the tomatoes again for about 20 seconds.
-
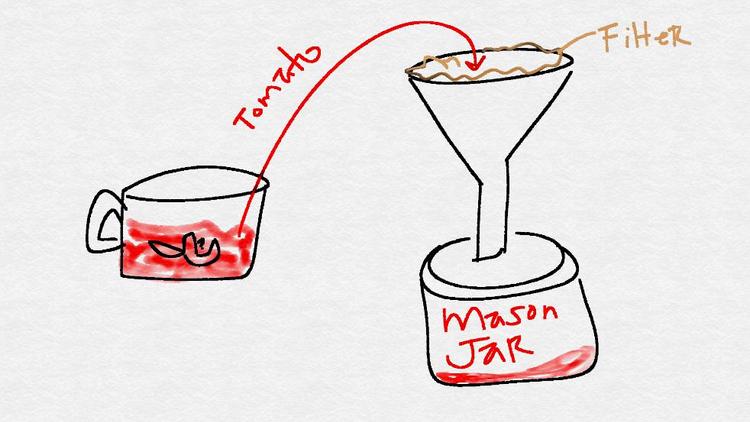
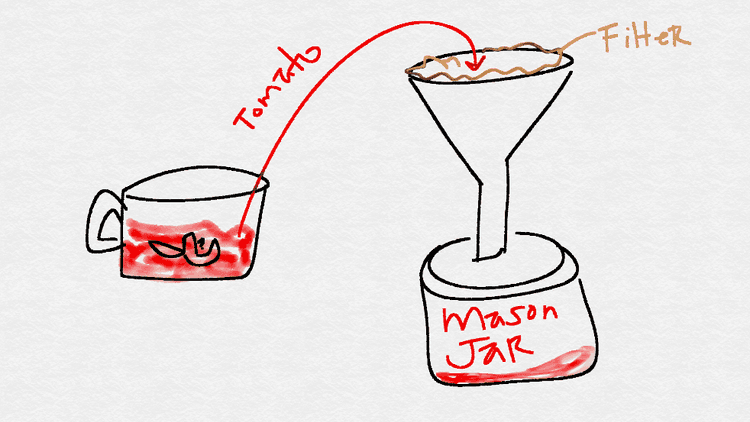
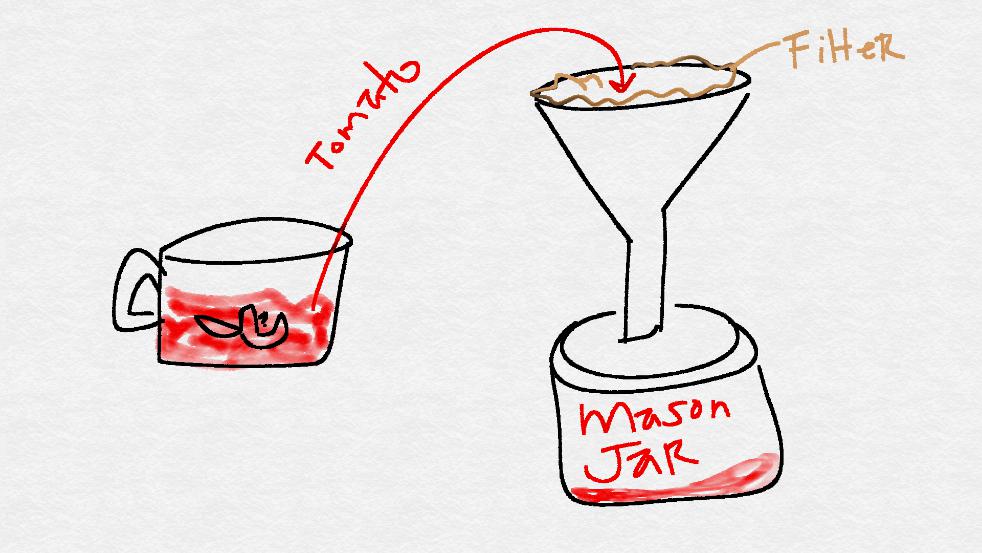
Strain the contents of the food processor into one of the mason jars using a coffee filter and funnel.
- Pour half of the mason jar's contents into another jar.
- Label the first jar as "tomato - raw."
-
Label the second jar as "tomato - cooked."

- Place the "tomato - cooked" jar on the hot plate at heat level 6 for 15 minutes. Do not allow the substance to boil.
- Repeat Steps 1 - 9 with the cherries.
-
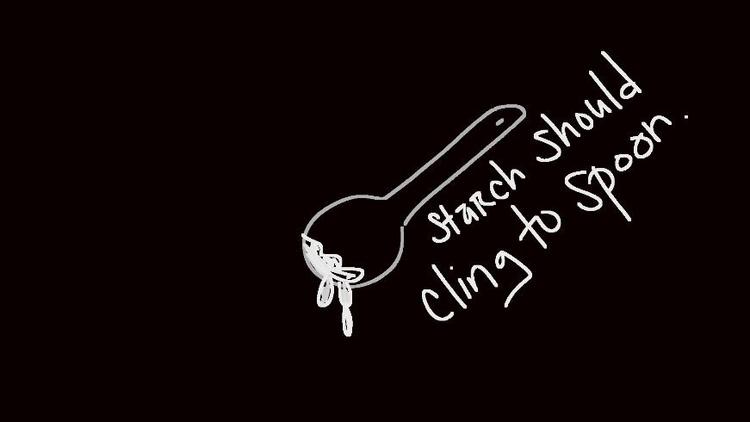
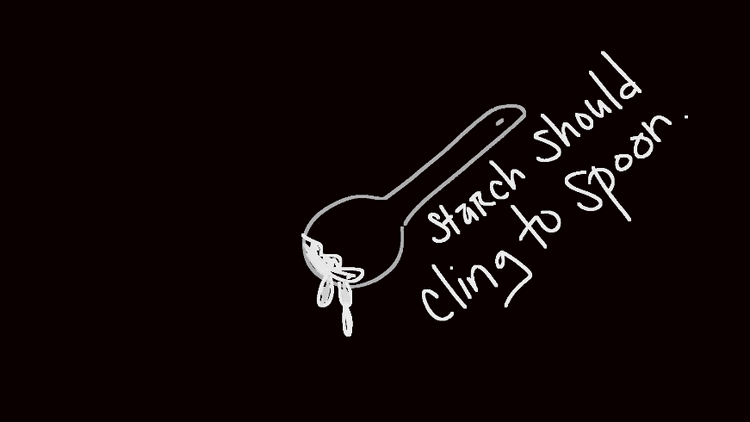
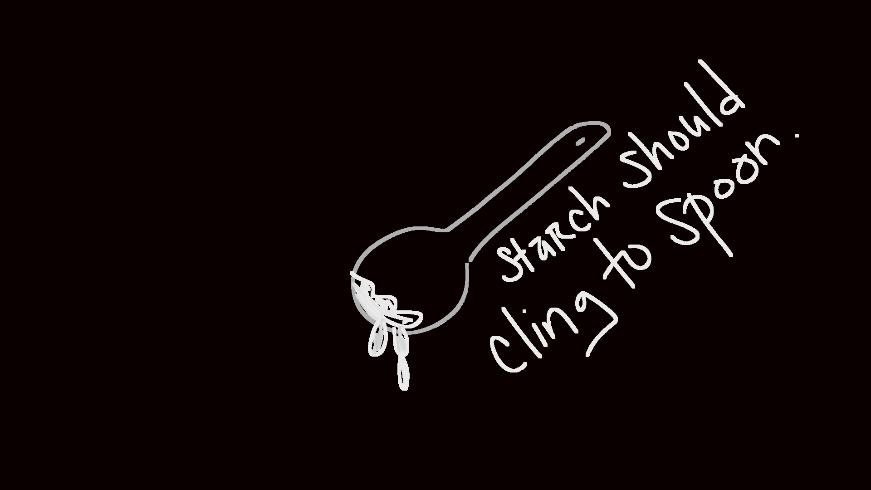
Add 1 tablespoon of cornstarch to another jar and add drops of water with one of the eyedroppers. Stir until it has a consistency of a paste.
- Measure 250 mL of water with the graduated cylinder.
- Add that water to your cornstarch mixture.
- Put the cornstarch mixture on the hot plate with heat at 8 ? 9 (when the starch boils). Boil for 5 minutes.
- Set the starch mixture aside to cool for about 5 minutes when done.
- Measure 75 mL of the distilled water into a clean mason jar.
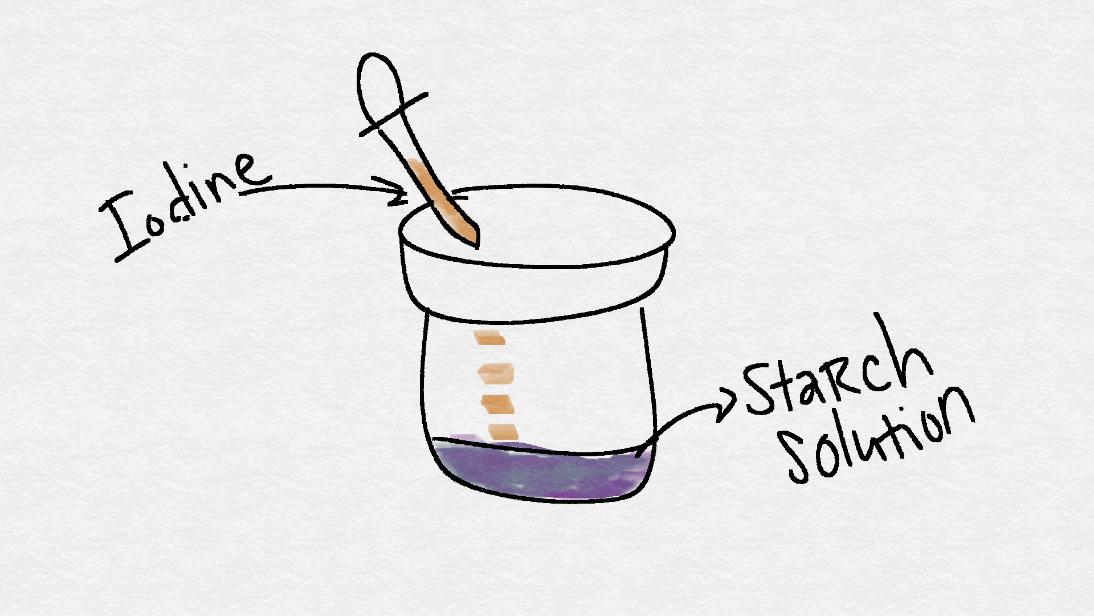
- Add 10 drops of the starch solution into the water using a clean eye dropper.
- Squeeze out any extra starch solution back into the jar it came from.
- Using a clean eye dropper, add the 2% iodine solution until it turns dark purple. Add it drop-by-drop.
- Repeat steps 17 - 20 in 3 more mason jars. You should have 4 jars with iodine solution inside.
-
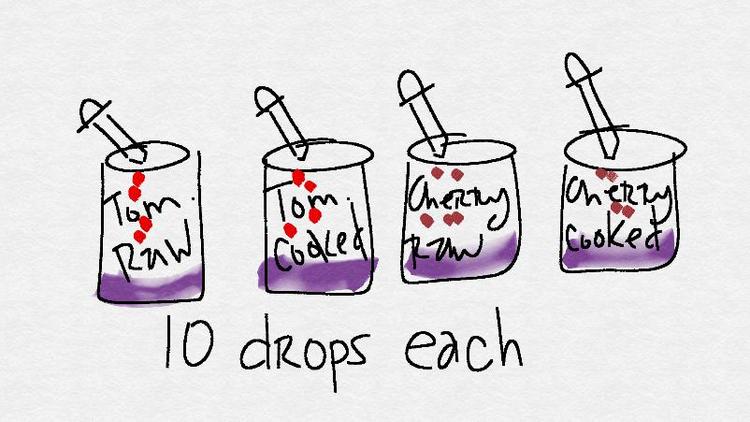
Label the jars with the extracts you intend to put inside. You should have jars with the following labels: "tomato - raw," "tomato - cooked", "cherry - raw," and "cherry - cooked."
- When the starch solution is a dark purple color, get a new eyedropper.
- Squeeze up some of the "tomato - raw" solution into your dropper. Add 10 drops.
-
Repeat the process by adding 10 drops of the "tomato - cooked", "cherry - raw," and "cherry - cooked" to the starch solution. Be sure that you are adding each extract to a fresh jar of iodine / starch solution.
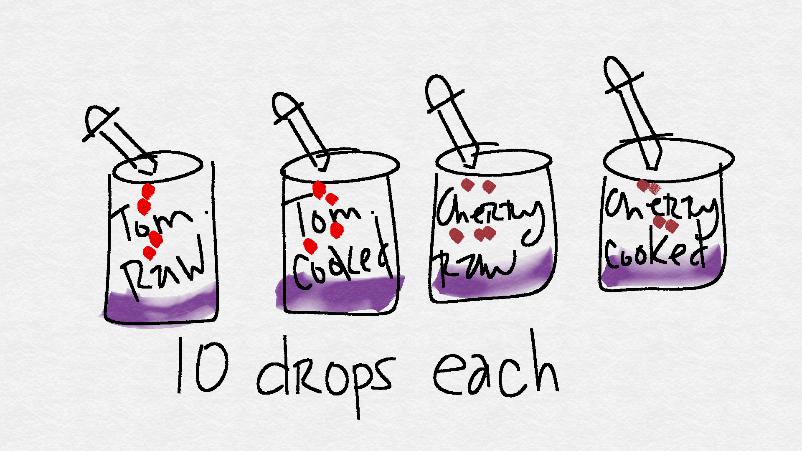
- When done, leave the jars next to each other for comparison and answer the discussion questions.